
Sign up for a new account.
And get access to
The latest T1D content
Research that matters
Our daily questions
Sign up by entering your info below.
Reset Your Password
Don't worry.
We will email you instructions to reset your
password.
T1D Exchange recently had the opportunity to interview Sean Salmon, President of Medtronic Diabetes. In this interview, we discuss Medtronic’s partnership with T1D Exchange and other entities to better understand and address the disparities that people of color living with diabetes face. We’ll also discuss Medtronic’s commitment to innovation in insulin delivery technology and access to these technologies for all people with diabetes.

Q1: Can you share a few details about your background, how long you’ve been with Medtronic and your current role, essentially overseeing Medtronic’s efforts and innovation in diabetes care?
A1: I have been at Medtronic for 17 years. I’ve been leading Medtronic Diabetes for the past two years where I’m responsible for the overall strategic direction and day-to-day global operations. Before this, I spent many years at Medtronic leading teams in the cardiovascular area. I’ve worked in the pharmaceutical and medical device industry for 30 years.
Q2: What thoughts do you keep top of mind about the needs of people with diabetes and their loved ones as you innovate to improve the lives of these individuals?
A2: Above all else, we want to make it easier to live with diabetes. Our goal is to find ways to make people think less about their diabetes — or even forget about it for some time. We approach this through technology innovations, but technology alone isn’t enough. We also want to make the overall experience of managing diabetes easier. At Medtronic we’re addressing this with our exceptional customer service and through new ways for customers to engage with us digitally such as supply ordering or product training.
![]() Q3: As noted Medtronic has partnered with T1D Exchange to better understand and address the disparities that people of color with diabetes face. What was the rationale for this partnership? Why is this work important for Medtronic?
Q3: As noted Medtronic has partnered with T1D Exchange to better understand and address the disparities that people of color with diabetes face. What was the rationale for this partnership? Why is this work important for Medtronic?
A3: When faced with data showing that people of color with diabetes are two-to-three times less likely to use diabetes technology, regardless of socioeconomic status, we believe that’s fundamentally wrong, and it’s our duty to do something about it. T1D Exchange is in a unique position to review data collected from in-person and telemedicine visits from more than 41 sites in the U.S. that are part of the Quality Improvement Collaborative (QIC). Using the QIC, clinicians can initially obtain a comprehensive baseline of technology use that is broken down by race and ethnicity. They can then longitudinally evaluate a series of interventions to determine their effect on technology adoption and utilization to improve many aspects of diabetes care. The community at-large can use these powerful insights to evolve care and thereby make a difference. We at Medtronic Diabetes we look forward to tackling this challenge with T1D Exchange.
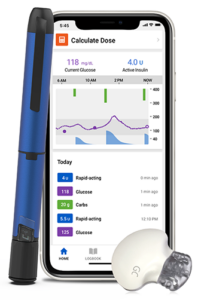 Q4: What is Medtronic’s commitment to innovation to make life easier, less burdensome, for people with T1D?
Q4: What is Medtronic’s commitment to innovation to make life easier, less burdensome, for people with T1D?
A4: Ultimately, we want to make living with diabetes simpler. We aim to reduce the mental burden of managing diabetes by focusing on technologies that automate insulin delivery or provide better decision support for the myriad decisions people with diabetes make daily. We’re also looking at reducing physical burden as well such as innovating an extended wear infusion set which can be worn for up to 7 days (versus up to 3 days for current sets).
It’s also important to us to meet each person with diabetes where they are in how they choose to manage their diabetes. Offering a variety of technology options enables the person to decide what is right for them at the time. This is why the addition of the InPen smart insulin pen system is so important to our portfolio of products. This smart pen makes it simple to track insulin doses, calculate doses and receive reminders and get support with other decisions that allows them to manage their diabetes more effectively without a lot of extra work.
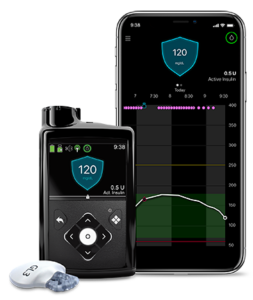 Q5: What new innovations and technologies can people with T1D and people with T2D who take insulin expect to see from Medtronic in the near future?
Q5: What new innovations and technologies can people with T1D and people with T2D who take insulin expect to see from Medtronic in the near future?
A5: We have our advanced hybrid closed loop insulin pump system available already in Europe and other parts of the world and look forward to getting more regulatory approvals to make it available to more people. This technology incorporates our advanced SmartGuard algorithm and will have the capability to connect with an updated CGM which is also currently under review by the FDA.
We’ll also be innovating on the InPen by using all that we’ve learned in creating our pump algorithms to “close the loop” on multiple daily injections (MDI). This will start by pairing the InPen with continuous glucose monitoring (CGM) to create a smart MDI system. We plan to develop dosing suggestion algorithms that will allow InPen users to also improve their glucose management without requiring a lot of extra work.
Q6: How will Medtronic help make your currently available and future insulin delivery technologies available to all people with diabetes, including people of color?
A6: Access to technology is critical for everyone. In addition to the work that we’re doing with T1D Exchange, we have a partnership with the American Diabetes Association (ADA) to support their Technology Access Project (TAP). The TAP aims to provide access to and availability of diabetes technology regardless of gender, race, income, or location. We also advocate with state and federal government to improve and expand access to diabetes technology. And, while we work to improve access, we are tackling affordability by making programs available to help anyone with financial need obtain the diabetes technology they need to live a healthy life.
Q7: What else do you think our readers would like to know about you and/or your and Medtronic’s commitment to people with diabetes?
A7: The most meaningful moments to me are when hear from people whose lives have changed because of Medtronic. In thinking about the intersection of innovation and access, I’m reminded of a young woman named Rukiyyah who was diagnosed with T1D during the Covid-19 pandemic. She started on MDI but quickly realized that she needed tighter glucose management and that a pump would better suit her active lifestyle. However, she had financial challenges which made getting this technology difficult. Our programs helped bridge this access gap and were able to get her onto the therapy she wanted. She says, “There are no limits anymore based on my diabetes.” That’s what we want for everyone.
Hope Warshaw
Related Stories
3 Comments
Get Up Close and in the Know with Sean Salmon, President of Medtronic Diabetes Cancel reply
You must be logged in to post a comment.








I am using the the Medtronic 770G pump and CGM. I generally like the system and my “time-in-rage” hovers around 80%. The improvements I would like to see most is in the CGM, the Sensor 3. The 7-day approved use is not long enough especially considering the cost of each sensor. It should be, at a minimum, 14 days if not 30. Many of us try to extend the sensor’s use and I sometimes get 14 days out of one. Also, while the sensor’s accuracy is good, it is not great. This could be improved. And why the 2 hour warm up for a newly inserted sensor? (When I try to extend the sensor a second week, I recharge the transmitter, reattach it, and again have a 2 hour warm up.)
I finally left Medtronic after waiting and waiting for them to get on track with quality control issues, and poor customer support. And I was with them for 20-ish years. What you cite about the sensor is right on. Dexcom’s accuracy blows Medtronic away. Tandem’s control IQ pump has also outperformed Medtronic.
Thank you for sharing your vision and mission for Medtronic. I’ve used a Medtronic insulin pump since 1999. Additionally, I have an implanted Medtronic pain pump I’ve used for 8 years. I recently purchased a Tandem pump, the T-Slim. Tandem uses a better quality and more accurate sensor; can be worn for 9 days and previously my Medtronic sensor required too many calibrations and the readings were 10-14 percent off when calibrating, on occasion even greater fluctuations. The new Medtronic pump is a huge leap forward and I would have stayed with Medtronic if they had used the Dexcom sensor or something comparable. I will miss Medtronic’s superior customer service. Medtronic is a great company that truly cares for their patients. Thank you for your fine leadership. Also, the smart pen device will be a game changer for many diabetics.