
Sign up for a new account.
And get access to
The latest T1D content
Research that matters
Our daily questions
Sign up by entering your info below.
Reset Your Password
Don't worry.
We will email you instructions to reset your
password.
The holy grail of insulin delivery for people with diabetes who use insulin is an automated insulin delivery system (AID) (preferred term for “artificial pancreas”). An AID system will deliver insulin through a so-called closed loop system that constantly maintains glucose levels within specified target range or ranges. That is, regardless of food consumed, physical activity, sleep, stress and myriad other factors that constantly impact glucose levels.
While the currently available AID systems, and those at the FDA for review or in development, inch their way towards this holy grail goal, they will not, in the near-future, truly and fully close the loop. Thus the term “hybrid-closed loop” is used.
Here we define AID systems and their components, draw attention to US Food and Drug Administration (FDA) review processes for these systems, and enumerate their benefits to users, caregivers and clinicians. You also find details about currently available AID systems in two tables and get a glimpse into the future. (Note: This article does not cover the Do-It-Your (DIY) systems (OpenAPS and Loop).
Current and Near-Future AID Systems
Over the last decade and with the brilliant expertise of and collaboration between dedicated experts in bioengineering, mathematics and the medical management of diabetes; steady progress has been made in the evolution towards AID systems. The availability of and advancement in continuous glucose monitoring (CGM) has made a critical impact on advancing this technology.
The evolution has followed a pathway from the Sensor-Augmented Pump (SAP) to Low Glucose Suspend (LGS) to Predictive Low Glucose Suspend (PLGS) to Hybrid Closed Loop (HCL) to Advanced Hybrid Closed Loop (AHCL) systems, which some refer to as AID systems.1 However, in reality they are partially closed being closed for basal insulin delivery but systems still requiring inputs for bolus insulin delivery. This evolution tracks the pathway mapped out by Aaron Kowalski, PhD, now JDRF’s Chief Executive Officer, in 20092 and updated in 2015.3
Today’s AID systems can increase basal insulin delivery to reduce hyperglycemia and suspend insulin delivery to reduce the risk and incidences of hypoglycemia.1 Most AID systems integrate an algorithm (defined below) that continuously utilizes CGM data to automatically correct glucose levels that are either above or below a specified target glucose range. Thus, these systems must integrate a CGM for data.
The reason that the currently available AID systems are considered “hybrid,” is because food intake, either by estimating carbohydrate consumption, inputting a meal announcement with relative meal size, or other method; must still be done and a bolus insulin dose given through the pump device by the user. For example, even with the Omnipod5, users will be advised to enter the amount of carbohydrate they consume in the device’s SmartBolus calculator, where the user will preset specified parameters for bolus dosing (see description in Table 1). A few studies on various AID systems have shown that when the user is in auto-mode/auto-correct some amount of elevated glucose that occurs due to not giving sufficient bolus insulin or missed food bolus insulin for a minimal amount of carbohydrate, can be covered.4,5,6
Three Main Automated Insulin Delivery Components
- CGM: A CGM device and its resulting data is an essential component. The CGM glucose results (data) are communicated to the device which are then used by the algorithm (defined below).
- Insulin delivery device: Today there are two types of delivery devices, one that requires the user to attach an infusion set (tubing) from the device to the skin. The other is a pod that attaches to the skin and delivers insulin thru a short, thin cannula. All continue to deliver insulin subcutaneously, as they always have.
- Algorithm: The algorithm can be considered the brains of an AID system that is built into the delivery device. There’s variation between the algorithms in AID systems. The various entities tend to be proprietary about these or use brand specific terminology to describe them.7 “An algorithm is a predetermined set of instructions used to tell the pump how to respond to the data (CGM) by providing or suspending insulin delivery,” says Davida Kruger, MSN, APN-BC, BC-ADM, certified adult nurse practitioner and diabetes clinician expert at Henry Ford Health System in Detroit. Kruger offers two examples: 1) the CGM reports to the pump the glucose level is 180 mg/dl. The algorithm assesses, based on the instructions, how much extra insulin a user needs and delivers it without the user taking any action. 2) The CGM provides data showing glucose is trending down in the next 30 minutes and if no change is made in the amount of insulin being delivered, the user will experience hypoglycemia. With this information, the pump automatically suspends insulin delivery and prevents hypoglycemia.
Bevy of Benefits
Anytime that people get relief from making one or many diabetes management decisions or performing daily tasks, it decreases disease burden. AID systems achieve both clinical and quality of life benefits.1
Clinically, research shows users of all ages can achieve a higher percent of glucose values within TiR (70 – 180 mg/dL)8 with less work. In hitting a higher TiR people hopefully minimize their Time Below Range (TBR), frequency of hypoglycemia and the need to treat hypoglycemia. Users also experience a lower percent of glucose results above TiRs or Time Above Range (TAR). In addition, studies show improvements in their Glucose Management Indicator (GMI)9 result and A1c.
AID researcher and person with T1D for 34 years Jennifer Sherr, MD, PhD, Associate Professor in Pediatrics (Endocrinology), Yale University School of Medicine, shares her observations. “With AID, users are finally able to hand over glycemic variability to algorithmically delivered insulin. This allows users to achieve more targeted A1c levels (goal of <7.0%). And of importance, AID technology accomplishes this while also offsetting disease burden. This has been especially notable in overnight hours and as studies have shown users experience improved sleep quality.”
Other quality of life benefits are less diabetes distress and relief of some daily management burdens, including all the mental math. In sum, people on AID systems find they have more time to focus on other aspects of their lives.
Review Processes of AID Systems
In the US, FDA’s Center for Diagnostics and Radiological Health (CDRH) reviews medical devices through one of several pathways. AID systems have generally been submitted as Class III medical devices which requires the entity seeking the review to submit a Premarket Approval Application (PMA). These applications include results of so-called pivotal studies required by FDA. Read A Look Inside the FDA’s Review Processes for Medical Devices (https://t1dexchange.org/a-look-inside-the-fda-approval-process-for-medical-devices/) to read how CDRH has revised review processes to allow for diabetes device innovation.
Around the globe entities work with other regulatory authorities. Entities may seek FDA review before doing so with other regulatory authorities or vise versa. For example, Medtronic’s MiniMed 780G is not yet approved in the US, but it has received CE mark in Europe and several other continents.
The AID Evolution Continues
AID systems will continue to evolve. The Kowalski pathway in Figure 1 offers insights as to how.3,4 Each advancement, such as eliminating the need announce food or carbohydrate intake or adding delivery of another hormone like glucagon10 or pramlintide,11 should make managing insulin-requiring diabetes a bit easier and close the loop further.
In the future you may see the availability of faster-acting insulins that require essentially no lead time before they quickly lower glucose as desired. You’ll likely see bi-hormonal AID systems as noted above,10,11 however, none are approved to date. In addition, there will likely be integration of decision support technologies that use artificial intelligence and machine learning, as noted in Table 1 under the description of Medtronic’s Personalized Closed Loop. Yes, step-by-step AID is advancing!
Sidebar: Glossary of Terms
New device categories give rise to new terms. That’s true with AID systems and changes made at FDA to revise review processes. The following are a few. For more details read: A Look Inside the FDA’s Review Processes for Medical Devices (https://t1dexchange.org/a-look-inside-the-fda-approval-process-for-medical-devices/).
- HCL (Hybrid Closed Loop): Currently available AID systems are considered “hybrid,” because they have not yet fully closed the loop. The user still must consider food intake, decide on a bolus dose and deliver it via the delivery device.
- AHCL (Advanced Hybrid Closed Loop): Similar to HCL, used by some entities to define their AID system.
- ACE pump: This FDA authorization opened the door to the first interoperable, automated glycemic controller device (iAGC) that automatically adjusts insulin delivery by connecting to an alternate controller-enabled insulin pump (abbreviated ACE pump) and integrated continuous glucose monitor (iCGM)
- iCGM: A CGM that is integrated with other compatible medical devices and electronic interfaces, such as automated insulin dosing systems, insulin pumps, BGM or other electronic devices used for diabetes management.
- Connectivity: The ability of diabetes management devices to communicate with each other, transfer diabetes-related data and provide this data in understandable, useable reports for user and clinician collaboration.
- Interoperability: The ability, potentially in the future, to purchase different device components (based on the user’s device preferences), and not to have to purchase the complete system from just one manufacturer.
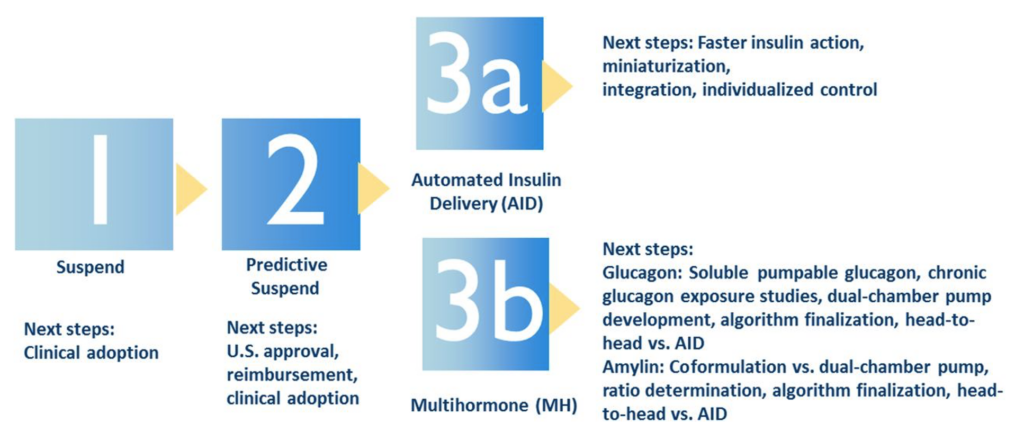 Figure 1: Kowalski A. Pathway to artificial pancreas systems revisited: Moving downstream. Diabetes Care. 2015;38:1036-1043. Reprinted with permission?
Figure 1: Kowalski A. Pathway to artificial pancreas systems revisited: Moving downstream. Diabetes Care. 2015;38:1036-1043. Reprinted with permission?
References:
- Grunberger G, Sherr J, Allende M, et al. American Association of Clinical Endocrinology clinical practice guideline: the use of advanced technology in the management of persons with diabetes mellitus. Endocrine Practice. 2021;27:505-537.
- Kowalski A. Can We Really Close the Loop and How Soon? Accelerating the Availability of an Artificial Pancreas: A Roadmap to Better Diabetes Outcomes. Diabetes Tech Therapeutics. 2009;Supp 1:S113-S119.
- Kowalski A. Pathway to artificial pancreas systems revisited: Moving downstream. Diabetes Care. 2015;38:1036-1043.
- Collyns OJ, Meier RA, Betts ZL, et al. Improved glycemic outcomes with Medtronic MiniMed advanced hybrid closed-loop delivery: results from a randomized crossover trial comparing automated insulin delivery with predictive low glucose suspend in people with type 1 diabetes. Diabetes Care. 2021;44(4):969-975.
- Bergenstal RM, Nimri R, Beck RW, et al. A comparison of two hybrid closed-loop systems in adolescents and young adults with type 1 diabetes (FLAIR): a multicentre, randomised, crossover trial. Lancet. 2021;397(10270):208-219.
- Sherr JL. Impact of the MINIMEDTM AHCL system on post-prandial glucose after a missed meal bolus in adolescents and adults with type 1 diabetes. 10th Annual Diabetes Technology Meeting. 2020.
- Messer LH, Forlenza GP. A clinical guide to advanced diabetes devices and closed-loop systems using the CARES paradigm. Diabetes Tech Therapeutics. 2019;21(8):462-469.
- Battelino T, Danna T, Bergental RM, et al. Clinical targets for continuous glucose monitoring data interpretation: recommendations from the international consensus on time in range. Diabetes Care. 2019;42(8):1593-1603.
- Bergenstal RM, Beck RW, Close KL, et al. Glucose management indicator (GMI): T new term for estimating A1C from continuous glucose monitoring. Diabetes Care. 2018;41(11):2275-2280.
- El-Khatib FH, et al. Home use of bi-hormonal bionic pancreas versus insulin pump therapy in adults with T1D: multicentre randomised crossover trial. Lancet. 2017;389:369-380.
- Tsoukas M, Cohen E, Legault L, et al. Alleviating carbohydrate counting with a FiASP-plus-pramlintide closed-loops delivery system (artificial pancreas): feasibility and pilot studies. Diabetes Obes. Metab. 2021;1-9.
Hope Warshaw
Related Stories
2 Comments
Status Update: Automated Insulin Delivery Devices Cancel reply
You must be logged in to post a comment.








Thx, Hope Warshaw! But your article references a table and a figure, although the web-visible version of your article has no HTML link indicated for either one.
Will our T1Dexchange subscriptions automatically inform us if your page gets updated?